Peptides and Cancer: The Truth Is More Complicated Than You’ve Been Told
Why some peptides may increase cancer risk, others may reduce it—and why NAD⁺ and Epitalon challenge the entire framework
Walk into any modern longevity clinic—or spend a few hours reading about peptides online—and you’ll quickly run into a contradiction.
Some people describe peptides as powerful tools for healing and healthy aging. Others warn that they may quietly increase cancer risk.
Both perspectives are grounded in real biology.
And both miss the bigger picture.
Peptides don’t have a single relationship with cancer. Some operate through pathways that tumors rely on. Others strengthen the systems that prevent cancer from forming in the first place. And a few—like NAD⁺ and Epitalon—sit squarely in the middle, forcing a more nuanced conversation.
What often gets lost is context. Compared to the dominant drivers of cancer—tobacco use, obesity and metabolic dysfunction, chronic inflammation, environmental and chemical exposures, poor diet, alcohol, radiation, infections, hormonal and IGF-1 signaling, aging, and genetics—the risk profile of most peptides is far more conditional and generally smaller in magnitude. They rarely act as primary causes. Instead, they tend to modify the biological environment in which cancer either develops or is suppressed.
To understand what’s actually going on, you have to stop thinking in terms of “safe” versus “dangerous” and start thinking in terms of mechanisms, timing, and biological context.

The Rule That Explains Almost Everything
Cancer is not foreign tissue. It’s your own cells behaving too well—growing, adapting, and surviving beyond normal limits.
That creates an uncomfortable overlap: many of the same systems that support healing and longevity are the ones cancer uses to thrive.
Which leads to a simple rule:
Anything that promotes growth can promote cancer—unless it strengthens the systems that eliminate cancer first.
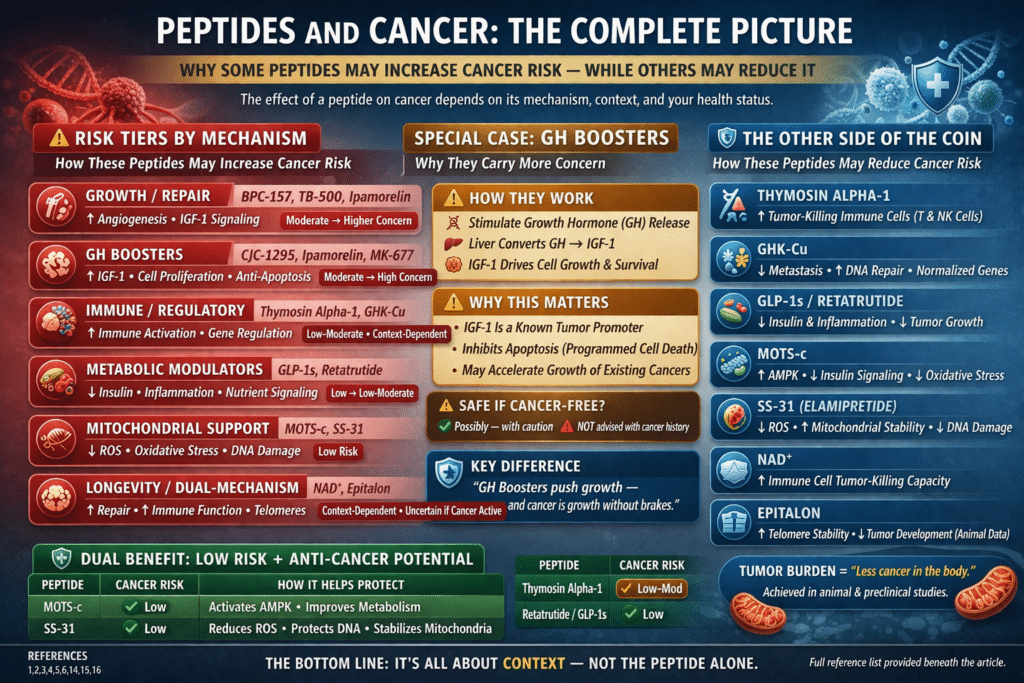
A Practical Framework: Risk Tiers by Mechanism
| Category | Peptides / Compounds | Cancer-Naive | Cancer History / Active | Mechanistic Direction |
|---|---|---|---|---|
| Growth / Repair | BPC-157, TB-500, Ipamorelin | Moderate | Higher Concern | Angiogenesis, IGF-1 signaling |
| Immune / Regulatory | Thymosin Alpha-1, GHK-Cu | Low–Moderate | Context-dependent | Immune activation, gene regulation |
| Metabolic Modulators | GLP-1s, Retatrutide | Low | Low–Moderate | Insulin, inflammation, nutrient signaling |
| Mitochondrial Support | MOTS-c, SS-31 | Low | Low | Mitochondrial efficiency, ROS reduction |
| Longevity / Dual-Mechanism | NAD⁺, Epitalon | Context-dependent | Higher Uncertainty | Energy metabolism, telomeres |
The Other Side of the Coin: Potential Anti-Cancer Directionality
| Category | Peptides / Compounds | Potential Anti-Cancer Effect | Mechanism |
|---|---|---|---|
| Growth / Repair | BPC-157, TB-500 | Tissue normalization (indirect) | Reduced chronic inflammation (limited evidence) |
| Immune / Regulatory | Thymosin Alpha-1 | Direct tumor suppression | Enhances T-cell and immune response³ |
| GHK-Cu | Reduced metastasis potential | Gene regulation, reduced invasion⁴⁵ | |
| Metabolic Modulators | GLP-1s, Retatrutide | Less cancer-permissive environment | Improved insulin sensitivity, reduced inflammation |
| Mitochondrial Support | MOTS-c | Anti-cancer metabolic signaling | AMPK activation, metabolic regulation¹⁵ |
| SS-31 | Reduced DNA damage risk | Lower ROS, mitochondrial stabilization¹⁶ | |
| Longevity / Dual-Mechanism | NAD⁺ | Enhanced immune surveillance | NK and CD8⁺ T-cell function⁸⁹ |
| Epitalon | Possible delayed tumor development | Telomere regulation¹⁰ |
When Healing and Cancer Use the Same Machinery
Some of the most popular peptides—like BPC-157 and TB-500—accelerate healing by promoting angiogenesis, the formation of new blood vessels. While beneficial for injured tissue, this same process is essential for tumor growth beyond a microscopic size.¹
Growth hormone–stimulating compounds such as ipamorelin raise a similar concern. By increasing IGF-1 signaling, they shift the body toward a more growth-permissive state—useful for recovery, but potentially problematic if malignant cells are already present.²
These compounds are not inherently dangerous, but they are aligned with growth biology, and that alignment matters.
GH Boosters and Cancer Risk
Growth hormone (GH) sits upstream of one of the most important signaling pathways in cancer biology: the insulin-like growth factor 1 (IGF-1) axis.
Physiologically, the pathway is straightforward:
- GH is released from the pituitary
- The liver responds by producing IGF-1
- IGF-1 drives cell proliferation, survival, and reduced apoptosis
Those same properties that make IGF-1 valuable for tissue repair also make it highly relevant to oncology.
Large epidemiological and mechanistic studies have consistently linked higher circulating IGF-1 levels with increased risk of several cancers, including breast, prostate, and colorectal malignancies.²¹³
Compounds that increase endogenous GH—such as:
- Ipamorelin
- CJC-1295
- MK-677 (ibutamoren)
therefore shift the body toward a more growth-permissive internal environment.
This does not mean they initiate cancer.
But it does mean they can influence two key processes central to tumor biology:
- Increased cellular proliferation²
- Reduced programmed cell death (apoptosis)²
In practical terms, this creates a context-dependent risk profile:
- In otherwise healthy individuals, especially with short-term use, the impact may be minimal
- In the presence of existing malignant or pre-malignant cells, elevated IGF-1 signaling may accelerate tumor growth
This aligns with broader clinical observations: individuals with chronically elevated GH/IGF-1 signaling—such as those with acromegaly—demonstrate increased rates of certain cancers.¹⁴
As with other growth-aligned peptides, the key distinction is not whether these compounds “cause” cancer, but whether they support the biological conditions that allow cancer to progress.
The Other Side: Strengthening the Body’s Defense System
If some peptides raise concern because they promote growth, others appear to strengthen the body’s ability to detect and eliminate cancer early.
Thymosin Alpha-1 is the clearest example, with established use as an immunomodulatory adjunct in oncology.³
More nuanced is GHK-Cu. Early concern centered on its role in tissue remodeling and angiogenesis. However, gene expression studies—primarily from a limited number of research groups—suggest a different pattern.
GHK has been reported to modulate dozens of genes involved in DNA repair and anti-cancer processes, and to reverse a substantial portion of metastatic gene expression signatures in colon cancer models.⁴ In breast and prostate cancer cell lines, it has been observed to reduce migration and invasion.⁴
Its angiogenic behavior also appears to be context-dependent, promoting vascularization in injured tissue while showing inhibitory effects on tumor-associated pathways in experimental cancer models.⁵
While these findings are largely preclinical and require broader independent replication, they suggest that GHK-Cu may function less as a simple growth signal and more as a regulatory peptide influencing cellular behavior.
Mitochondrial Peptides: A Different Axis
MOTS-c
LOW RISK — Both Populations
MOTS-c is a mitochondria-encoded peptide that regulates metabolic homeostasis, improves insulin sensitivity, reduces oxidative stress, and activates AMPK signaling — a pathway associated with longevity and metabolic health. No oncogenic signaling mechanisms have been identified in published research.¹⁵
- Improves insulin sensitivity
- Regulates fat metabolism
- Enhances mitochondrial efficiency
Its metabolic effects — insulin sensitization, fat metabolism regulation, mitochondrial efficiency — are directionally anti-cancer rather than pro-cancer, as metabolic dysfunction and hyperinsulinemia are established cancer risk factors. No studies have raised specific cancer concerns for MOTS-c.
Bottom line: One of the more favorable risk profiles on this list. The metabolic mechanism is directionally protective. Suitable for both populations with standard precautions.
SS-31 (Elamipretide)
LOW RISK — Both Populations
SS-31 works by stabilizing cardiolipin on the inner mitochondrial membrane, reducing reactive oxygen species, and restoring mitochondrial energy production. Oxidative stress and mitochondrial dysfunction are contributors to carcinogenesis — making SS-31’s mechanism directionally protective.¹⁶
- Reduces reactive oxygen species (ROS)
- Stabilizes mitochondrial membranes
- Restores energy production
No proliferative signaling pathways, angiogenic effects, or oncogenic mechanisms have been identified for SS-31. Clinical trials in heart failure and mitochondrial myopathy, though they did not meet primary endpoints, produced no oncological safety signals.¹⁷
Bottom line: Low cancer concern for both populations. The mitochondrial protection mechanism is theoretically anti-carcinogenic, and the clinical trial experience provides reassurance on basic safety.
Metabolism and Tumor Biology: The Retatrutide Signal
One of the strongest drivers of cancer risk is not any individual peptide, but the underlying metabolic environment in which tumors develop.
This is where newer multi-agonist therapies like retatrutide become particularly interesting—not just for weight loss, but for their effects on tumor biology.
In preclinical models of obesity-associated cancers, GLP-1–based and related incretin therapies have been shown to influence tumor growth dynamics alongside improvements in systemic metabolic dysfunction.⁶
These effects appear to be mediated through several converging mechanisms:
- Reduction in hyperinsulinemia, a known tumor growth signal
- Decreases in chronic inflammation
- Modulation of nutrient-sensing pathways that cancer cells rely on
In these models, such changes translated into slower tumor growth and reduced tumor burden compared to untreated controls.⁶
Importantly, these findings remain preclinical, and human cancer-specific outcome data is not yet available.
Still, the implication is significant:
Altering the metabolic environment may influence cancer not by targeting the tumor directly, but by removing the conditions that allow it to thrive.
NAD⁺: The First Paradox
NAD⁺ has long been viewed with caution in oncology due to its central role in cellular energy metabolism.
Tumor cells depend heavily on NAD⁺ to sustain growth and manage oxidative stress.⁷
At the same time, NAD⁺ is critical for immune function. Restoration of NAD⁺ levels has been shown to enhance natural killer cell cytotoxicity and improve CD8+ T cell metabolic fitness and tumor-killing capacity.⁸⁹
Within tumor environments, NAD⁺ depletion is associated with impaired immune response, while replenishment can restore anti-tumor activity.⁸
This creates a dual reality:
- NAD⁺ can support tumor metabolism
- NAD⁺ increases immune tumor-killing capacity by strengthening NK cell and CD8⁺ T cell function
Which effect dominates depends largely on biological context.
Epitalon: The Deeper Paradox
If NAD⁺ complicates metabolism, Epitalon complicates something even more fundamental: cellular aging itself.
Epitalon is a pineal-derived tetrapeptide that activates telomerase, the enzyme responsible for maintaining telomere length.¹⁰
Because telomerase activation is a hallmark of many cancers, this mechanism initially raised concern.
But the data does not behave the way that concern would predict.
In normal human cells, Epitalon has been shown to increase telomerase activity and extend telomeres.¹¹ In cancer cell models, however, it appears to activate alternative telomere maintenance pathways (ALT) rather than directly increasing telomerase activity—suggesting that its effects may differ depending on cellular context.¹¹
Animal studies further complicate the picture. Rather than increasing cancer incidence, Epitalon has been associated with delayed tumor development and, in some cases, reduced tumor burden, including reductions in leukemia incidence in certain models.¹⁰
The most provocative data, however, comes from small human cohort studies conducted over multi-year periods.
In one widely cited study of 266 elderly individuals followed for 6–8 years, the control group experienced an 81.8% mortality rate, while those receiving Epithalamin showed markedly lower mortality:
- Epithalamin alone: 41.7%
- Thymalin alone: 45.8%
- Combined therapy: 33.3%
- Combined with repeated annual treatment: 20.0%¹²
In practical terms, the most intensively treated group experienced roughly a 4-fold reduction in mortality compared to controls.
Additional observations have reported:
- 1.6–1.8× reductions in overall mortality
- Up to 2.5× reductions when combined with thymic peptides
- Improvements in cardiovascular and metabolic function¹²
This creates a difficult tension:
If Epitalon were meaningfully increasing cancer-related mortality, it would be hard to reconcile that with large reductions in all-cause mortality over multi-year periods.
At the same time, these findings must be interpreted cautiously. The studies involve relatively small cohorts, lack modern randomized controls, and have been produced largely within a single research network with limited independent replication.
The result is a compound that is:
- Biologically plausible
- Clinically intriguing
- Still genuinely unresolved
Why the Uncertainty Still Matters
Despite intriguing signals, Epitalon research remains concentrated within a relatively small group of investigators, with limited independent validation.
That leaves us in an unusual position:
- The observed effects are substantial
- The level of external replication is limited
Until larger, independently conducted studies confirm these findings, Epitalon remains scientifically promising—but not yet clinically established.

Context Still Decides Everything
Just as with NAD⁺, Epitalon’s risk profile depends heavily on context.
- In healthy individuals, current evidence does not demonstrate increased cancer incidence and may suggest protective or longevity-related effects
- In individuals with active cancer, recent remission, or high genetic risk, the involvement of telomerase biology introduces enough uncertainty to warrant caution
The Bigger Picture
Peptides are not inherently “good” or “bad” for cancer.
They are biological levers, acting on:
- Growth
- Immune surveillance
- Metabolism
- Cellular aging
Some push in one direction. Others push in the opposite direction. And a few—like NAD⁺ and Epitalon—act in both.
Final Thought
The most useful question is no longer:
“Does this peptide increase cancer risk?”
It’s:
“What system is this peptide influencing—and what condition is my body in when I use it?”
Because in the end, the difference between protection and risk is rarely the molecule alone.
It’s the context in which it acts.
References
- Carmeliet P, Jain RK. Angiogenesis in cancer and other diseases. Nature. 2000;407(6801):249–257.
- Pollak M. Insulin and insulin-like growth factor signalling in neoplasia. Nat Rev Cancer. 2008;8(12):915–928.
- Costantini C, Bellet MM, Pariano M, et al. Thymosin alpha-1 as a regulator of immunity in cancer therapy. Front Oncol. 2019;9:873.
- Pickart L, Margolina A. Regenerative and protective actions of the GHK-Cu peptide in the light of the new gene data. OBM Genet. 2021;5(2):128.
- Pickart L. The human tri-peptide GHK and tissue remodeling. J Biomater Sci Polym Ed. 2008;19(8):969–988.
- Drucker DJ. Mechanisms of action and therapeutic application of glucagon-like peptide-1. Cell Metab. 2018;27(4):740–756.
- Chiarugi A, Dölle C, Felici R, Ziegler M. The NAD metabolome—a key determinant of cancer cell biology. Nat Rev Cancer. 2012;12(11):741–752.
- Morandi F, Horenstein AL, Costa F, et al. NAD⁺ metabolism and cancer immunotherapy. Front Immunol. 2021;12:647590.
- Xiao Y, Chen X, Chen W, et al. NAD⁺ metabolism and CD8⁺ T cell function in cancer. Nutrients. 2024;16(20):3577.
- Khavinson VKh, Linkova NS, Dyatlova AS, et al. Peptide regulation of gene expression and aging. Int J Mol Sci. 2020;21(20):7460.
- Al-Dulaimi S, et al. Epitalon and telomere biology: a review. Biogerontology. 2021;22(1):1–12.
- Khavinson VKh, Morozov VG. Pineal peptides and aging outcomes. Neuroendocrinol Lett. 2003;24(1–2):67–72.
- Renehan AG, Zwahlen M, Minder C, et al. Insulin-like growth factor (IGF-1) and cancer risk: systematic review and meta-analysis. Lancet. 2004;363(9418):1346–1353.
- Colao A, Ferone D, Marzullo P, Lombardi G. Systemic complications of acromegaly: epidemiology, pathogenesis, and management. Endocr Rev. 2004;25(1):102–152.
- Lee C, Zeng J, Drew BG, et al. The mitochondrial-derived peptide MOTS-c promotes metabolic homeostasis. Cell Metab. 2015;21(3):443–454.
- Szeto HH. First-in-class cardiolipin-protective compound as a therapeutic agent to restore mitochondrial bioenergetics. Pharm Res. 2014;31(10):2619–2627.
- Daubert MA, Yow E, Dunn G, et al. Novel mitochondria-targeting peptide in heart failure treatment. Circ Heart Fail. 2017;10(12):e004389.