Peptides and Immunomodulation: From Chaos to Control
Immunomodulation, Cytokine Dysregulation, and the Role of Peptides in Chronic Inflammation
Executive Summary
Many chronic inflammatory and autoimmune conditions—including colitis, Hashimoto’s thyroiditis, endometriosis, and related disorders—are not caused by an immune system that is simply “too strong.” Instead, they reflect an immune system that has lost its ability to regulate itself.
This article explores immune dysfunction as a spectrum, ranging from acute cytokine storms to chronic low-grade inflammation, and explains why immune signaling can become trapped in maladaptive patterns over time. It introduces immunomodulation as a distinct biological strategy—focused on restoring immune balance rather than suppressing immunity—and examines how peptides such as Thymosin Alpha-1, along with supportive peptides like KPV, BPC-157, and combination strategies such as KLOW, may help guide the immune system back toward stability.
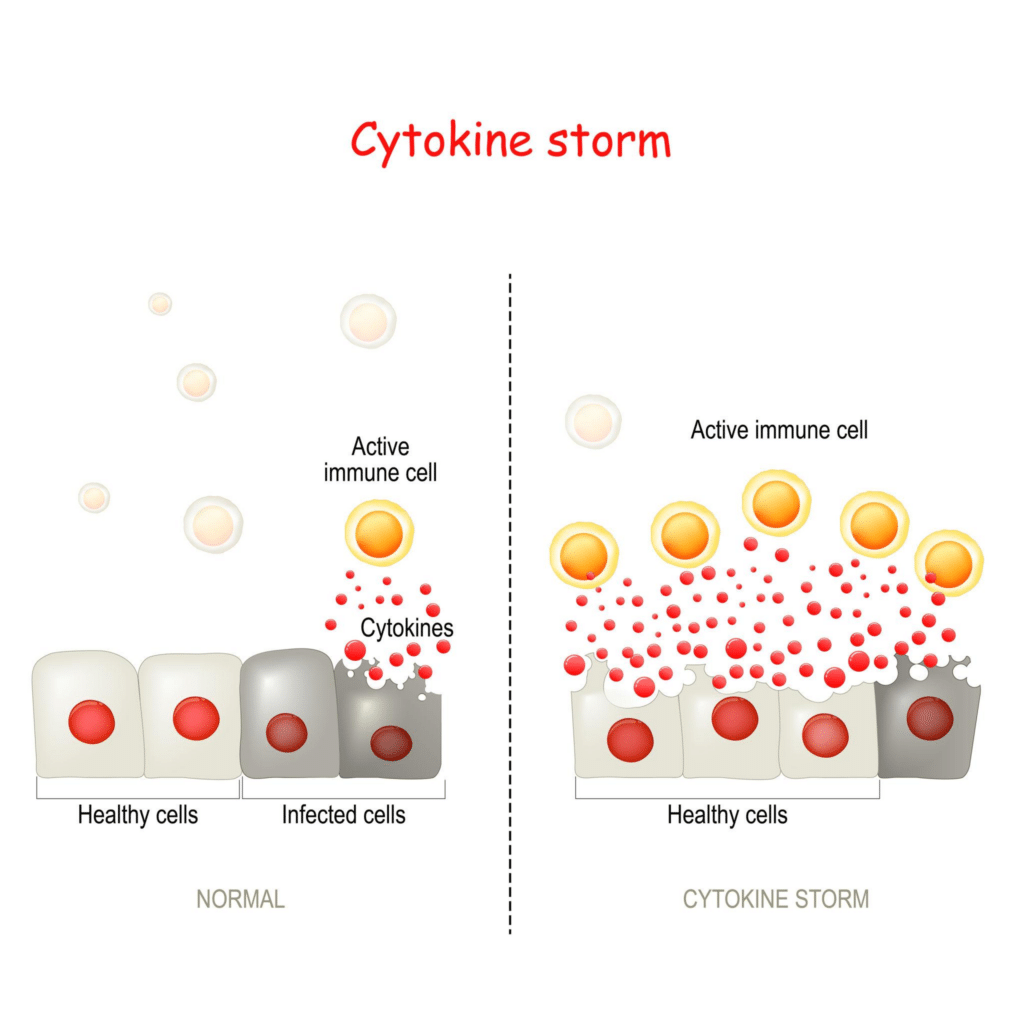
When Inflammation Becomes the Problem
For individuals living with autoimmune or chronic inflammatory conditions, symptoms often feel unpredictable. Long periods of relative stability may be interrupted by sudden flares that disrupt daily life, energy, and function. While these conditions affect different organs—the gut, thyroid, or pelvic tissues—the underlying biological pattern is strikingly similar.
In a healthy immune system, inflammation is a temporary tool. It turns on in response to a threat and turns off once that threat is resolved. In chronic disease states, this resolution step begins to fail. The immune system remains partially activated even in the absence of danger, creating a cycle in which inflammation itself becomes the primary driver of tissue damage and symptoms.1
The Spectrum of Immune Dysregulation
Immune dysfunction exists along a continuum rather than as a single uniform state. At one end lies the cytokine storm, an acute and uncontrolled release of pro-inflammatory cytokines such as interleukin-6 and tumor necrosis factor-alpha. This phenomenon is best known in severe infections but shares mechanistic overlap with intense autoimmune or inflammatory flares, including severe colitis exacerbations.2 In this state, inflammatory signaling fails to shut down, leading to rapid tissue injury and systemic exhaustion.
Further along the spectrum is cytokine dysregulation. In conditions such as Hashimoto’s thyroiditis, inflammation may not be explosive, but it is poorly resolved. Pro- and anti-inflammatory signals fall out of sync, leaving the immune system locked in a persistent state of alert. This represents not immune excess, but a failure of immune signal resolution.3
At the chronic end of the spectrum lies low-grade inflammation. Disorders such as endometriosis often fit this pattern. Here, immune cells remain persistently active, producing inflammatory mediators that promote pain, fibrosis, and tissue remodeling over years rather than days.4 This slow, smoldering inflammation can be just as damaging as acute flares when sustained long term.
Why Immune Dysregulation Develops Over Time
Immune dysregulation rarely appears suddenly. In most individuals, immune responses earlier in life were appropriately calibrated—activating when needed and resolving once the threat had passed. Over time, however, several converging factors reduce the immune system’s capacity to self-regulate.
One of the most important contributors is age-related immune remodeling. The thymus, which plays a central role in training T lymphocytes, gradually involutes with age. This process reduces the output of naïve and regulatory T cells while skewing immune balance toward memory and effector populations.5 The immune system does not simply become weaker; it becomes less precise and less capable of resolution.
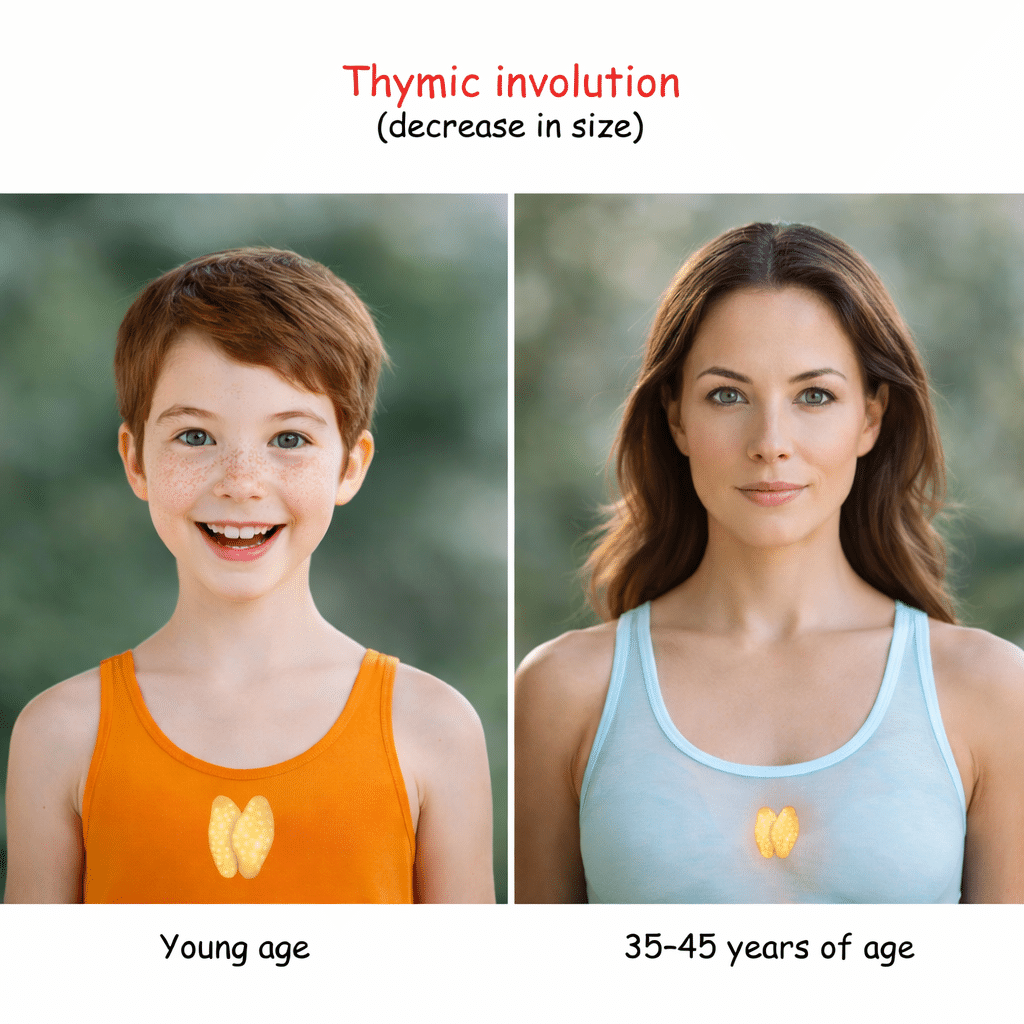
Chronic stress accelerates this decline. Psychological stress, metabolic strain, sleep disruption, and sustained physical stress all increase inflammatory signaling while impairing immune recovery. Elevated stress hormones alter cytokine production and disrupt feedback loops that normally terminate immune responses. Over time, this biases the immune system toward persistent activation.6
Environmental exposure compounds this burden. Repeated exposure to dietary antigens, toxins, endocrine disruptors, and microbial stimuli creates a constant low-level immune workload. Younger immune systems often absorb this burden without consequence. As immune reserve diminishes, however, repeated activation without adequate resolution leads to signaling fatigue and dysregulation.7
Infections may act as inflection points. Certain viral and bacterial exposures leave long-lasting imprints on immune signaling, maintaining elevated inflammatory tone even after clinical recovery. In genetically susceptible individuals, these residual immune changes may increase the risk of autoimmune or chronic inflammatory disease years later.8
Hormonal shifts further influence immune calibration. Changes in thyroid hormones, sex hormones, and cortisol alter immune signaling thresholds. These transitions often occur gradually with age or during periods such as postpartum recovery or perimenopause, increasing the likelihood of immune misinterpretation of internal signals as threats.9
Gut integrity also plays a central role. A large proportion of immune activity occurs within the gastrointestinal tract, where tolerance and defense must be carefully balanced. Disruption of the gut barrier—through inflammation, infection, medication use, or dietary factors—allows persistent immune stimulation that reinforces systemic cytokine dysregulation.10
Taken together, these influences create cumulative immune load. Over time, the immune system adapts by remaining partially activated, even when it should stand down. Immune dysregulation is therefore not a sudden failure, but the predictable outcome of long-term strain on immune regulatory capacity.
Immunomodulation vs Immune Suppression
Traditional anti-inflammatory strategies often focus on blocking immune pathways or suppressing immune activity altogether. While this can reduce symptoms, it may also impair normal immune defense and does not necessarily restore healthy immune coordination.
Immunomodulation takes a different approach. Rather than silencing immune responses, it aims to restore proportional signaling—allowing inflammation to occur when appropriate and resolve when its function is complete. This upstream focus on immune regulation is central to understanding the role of Thymosin Alpha-1.
How Thymosin Alpha-1 Restores Immune Balance
Thymosin Alpha-1 is a naturally occurring peptide derived from thymic tissue and plays a role in immune signaling and coordination. It is best understood as an immune training and calibration molecule rather than a direct anti-inflammatory agent.11
One of its primary actions involves supporting regulatory T cells, which serve as the immune system’s internal braking mechanism. These cells are essential for terminating inflammatory responses once threats are neutralized. In autoimmune flares and cytokine dysregulation, regulatory signaling is often impaired. Thymosin Alpha-1 has been shown to enhance regulatory T-cell function, helping restore immune resolution.12
The peptide also modulates inflammatory signaling pathways such as NF-κB. Rather than fully inhibiting these pathways, Thymosin Alpha-1 appears to influence their activation dynamics, reducing background inflammatory tone while preserving host defense.13
Additionally, Thymosin Alpha-1 supports immune surveillance and cleanup by improving dendritic cell and macrophage function. In chronic inflammation, these cells may perpetuate inflammatory signaling rather than resolve it. By promoting appropriate immune differentiation, Thymosin Alpha-1 helps shift immune activity from persistence toward repair.14
Repair After Regulation: Supporting Tissue Healing
Once immune signaling begins to normalize, tissue repair becomes the next priority. Chronic inflammation often leaves behind damaged barriers, impaired blood flow, and fibrotic remodeling.
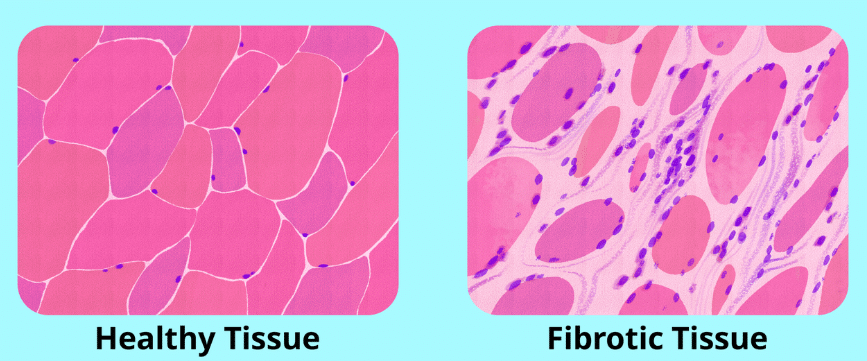
KPV is particularly relevant for localized inflammatory environments, where it helps reduce residual cytokine signaling at the tissue level.15 BPC-157 supports angiogenesis and epithelial repair, contributing to restoration of tissue integrity.16 Thymosin Beta-4 enhances cell migration, supporting coordinated repair across damaged tissues.17 GHK-Cu influences collagen organization and extracellular matrix remodeling, helping reduce excessive fibrosis and promote healthier tissue architecture.18
When a Broader Approach Is Needed: The KLOW Strategy
The KLOW combination—KPV, BPC-157, Thymosin Beta-4, and GHK-Cu—represents a systemic repair-oriented strategy. It is most appropriate in complex, multi-system cases where chronic low-grade inflammation has persisted long enough to affect multiple tissues simultaneously.
In earlier or more localized conditions, simpler peptide pairings layered on top of immune modulation may be sufficient. Escalation to broader strategies is best reserved for situations in which immune dysregulation and tissue damage have become tightly intertwined.
Restoring Harmony Rather Than Silencing the System
Chronic inflammatory and autoimmune conditions are rarely the result of an immune system that is simply overactive. More often, they reflect a system that has lost coordination, resolution, and regulatory feedback.
Immunomodulation reframes the goal of intervention: not to suppress immune activity, but to restore balance. Within this framework, peptides such as Thymosin Alpha-1 and its supportive counterparts function as biological signals—guiding the immune system back toward harmony rather than forcing it into silence.
References
- Medzhitov R. Origin and physiological roles of inflammation. Nature. 2008;454(7203):428-435.
- Tisoncik JR, et al. Into the eye of the cytokine storm. Microbiol Mol Biol Rev. 2012;76(1):16-32.
- Rose NR. Negative selection, epitope mimicry and autoimmunity. Curr Opin Immunol. 2017;49:51-55.
- Bulun SE. Endometriosis. N Engl J Med. 2009;360(3):268-279.
- Palmer DB. The effect of age on thymic function. Front Immunol. 2013;4:316.
- Dhabhar FS. Effects of stress on immune function. Annu Rev Psychol. 2014;65:205-231.
- Miller GW, et al. Environmental influences on immune dysfunction. Immunol Rev. 2012;245(1):168-186.
- Smatti MK, et al. Viruses and autoimmunity. Clin Immunol. 2019;203:99-111.
- Straub RH. The complex role of hormones in inflammation. Endocr Rev. 2007;28(5):521-574.
- Camilleri M. Leaky gut: mechanisms and implications. Gut. 2019;68(8):1516-1526.
- Garaci E, et al. Thymosin alpha 1: mechanism of action. Ann N Y Acad Sci. 2012;1270:19-26.
- Romani L, et al. Thymosin alpha 1 activates regulatory T cells. Blood. 2006;108(7):2269-2276.
- Goldstein AL, et al. Immunoregulatory properties of thymosin alpha 1. J Biol Regul Homeost Agents. 2014;28(3):357-367.
- King R, et al. Thymosin alpha 1 and dendritic cell function. Clin Immunol. 2015;160(2):199-207.
- Brzoska T, et al. Anti-inflammatory properties of KPV. J Invest Dermatol. 2008;128(1):214-223.
- Sikiric P, et al. BPC-157 and tissue healing. Curr Pharm Des. 2011;17(16):1612-1632.
- Goldstein AL, et al. Thymosin beta-4 in tissue repair. Ann N Y Acad Sci. 2012;1269:1-9.
- Pickart L. The human tri-peptide GHK and tissue remodeling. Biomed Res Int. 2015;2015:648108.